Pharmaceuticals & Life Sciences
We help quality-sensitive teams structure workflows, expose the right production context, and make data easier to review where traceability and consistency matter most.
Industry overview
Support controlled processes with clearer quality and data discipline
Controlled digital workflows, traceability-aware tools, and quality-focused visibility for regulated and process-sensitive production environments.
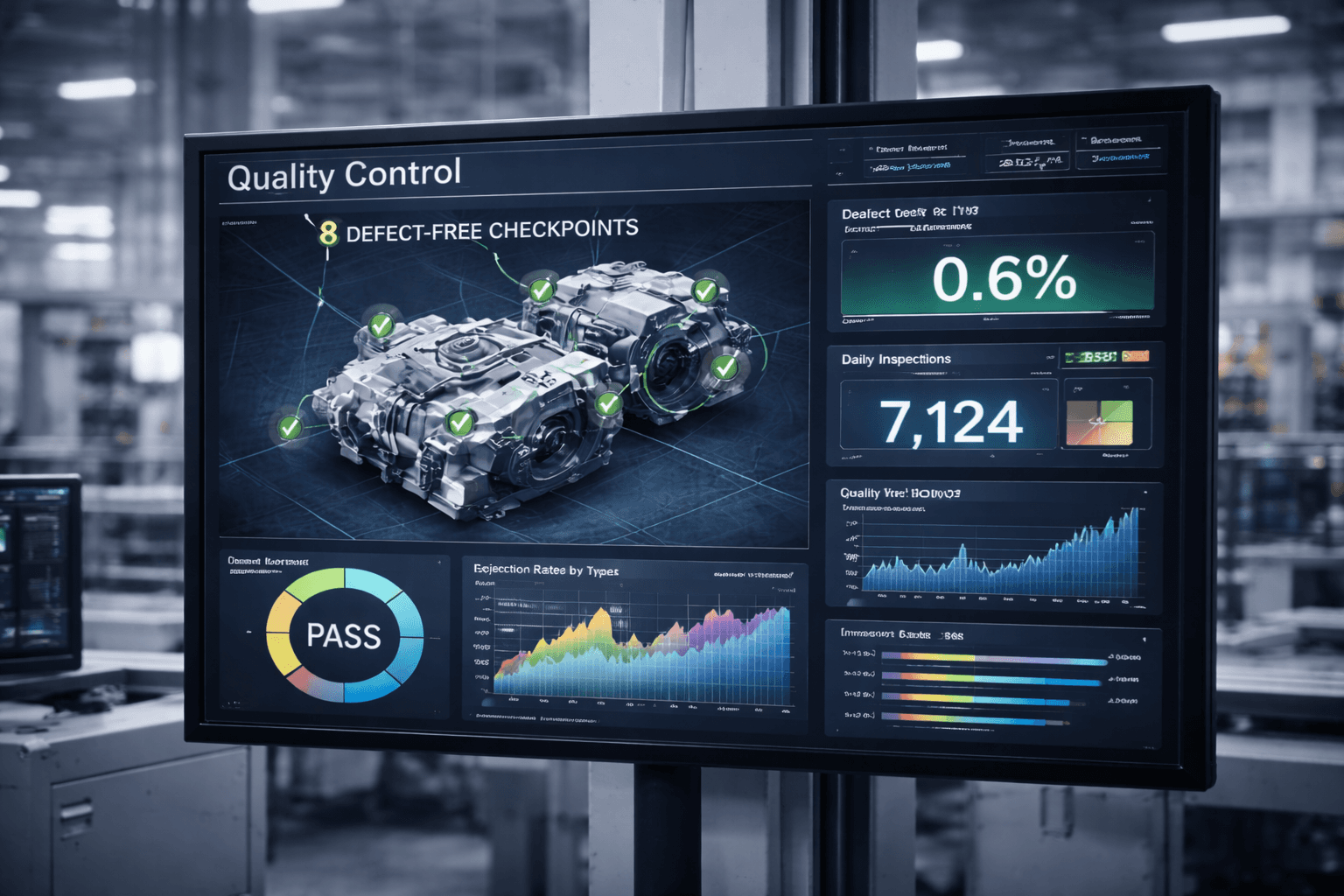
Pharmaceutical and life science environments place higher demands on consistency, documentation, and reliable process interpretation. The digital layer needs to support controlled execution and review, not simply display more data. That is where structured interfaces and traceability-aware design become especially valuable.
Production environment
Quality-sensitive workflows, batch or recipe context, controlled process steps, and inspection-heavy execution
Main priorities
Traceability, documentation quality, controlled workflows, and dependable data integrity in daily use
Typical stack
Structured storage, role-based tools, review-oriented interfaces, and quality process visibility layers
Workflow control and guidance
Digital steps and interface logic can help keep execution aligned with the intended process and reduce ambiguity in daily operation.
Traceability-aware data handling
Data is stored and presented with enough context to support quality review, investigation, and follow-up.
Inspection and quality visibility
Teams can review process and quality information in a structure that supports consistency rather than raw data overload.
Stronger process consistency
Clearer quality review context
Better traceability support
More disciplined data handling
